Take home messages
- Resistance exists in red legged earth mite (RLEM), green peach aphid (GPA), diamond back moth (DBM) and Corn Earworm (H. armigera) in the southern region.
- Resistance can occur from intentional (targeted pest) or unintentional (non-targeted pest) use of the same mode of action (MoA) repeatedly.
- Because all insecticides within a group share a common MoA, there is a high risk of cross‑resistance to many insecticides in the same group.
- In broadacre farming, rotating use of the commonly used synthetic pyrethroid (group 3A) and organophosphate groups (1B) with other groups (where possible), will help to minimise evolution of resistance in target and non-target pests.
What is insecticide resistance?
Insecticide resistance has been documented as far back as 1914, as reported by Axel Melander in a research article entitled ‘Can insects become resistant to sprays?’. By the mid 1940’s 11 cases of resistance to inorganic insecticides had been documented, and with the introduction of the first organic insecticide to the market in 1942 (DDT) it was not long before housefly resistance was documented in 1947. Since that time, resistance to organic synthetic insecticides have been reported at an increasing rate for many chemicals introduced to the market.
This is known as the insecticide treadmill, and due to the nature of genes and mutation, selection of beneficial traits, and often short invertebrate generation times, this trend is likely to continue in the absence of significant practice change. Globally, there are more than 580 documented cases of invertebrate pests evolving resistance, and 325 unique chemicals for which one or more species have evolved resistance.
The Australian grains industry has an ongoing reliance on limited chemical control methods – particularly the cheaper broad-spectrum chemistries. Therefore, managing resistance is a major challenge. Currently insecticide resistance in Australian grains (excluding the grain storage pests) is established in redlegged earth mites (Halotydeus destructor), green peach aphid (Myzus persicae), diamondback moth (Plutella xylostella) and corn earworm (Helicoverpa armigera) (Table 1).
How does resistance evolve?
All insecticides belong to a particular ‘Mode of Action’ (MoA) group. Each MoA group results in the same functional or anatomical change in an invertebrate at the level of the cell. The MoA group is listed on the chemical label. Resistance occurs when repeated applications of a MoA insecticide group removes susceptible insects from successive populations, leaving increasing numbers of individuals that are resistant.
Under selective pressure from insecticides, resistant individuals are more genetically ‘fit’ than their susceptible counterparts. Mating between these resistant individuals gradually increases the proportion that are resistant in the pest population. Eventually this can render an insecticide ineffective, leading to control failures in the field. Resistance can arise due to selection for:
1. A heritable trait already present in a small portion of the pest population that provides resistance,
or
2. A completely new heritable trait (a mutation) in an individual or individuals that provide resistance.
The main mechanisms of resistance are target site, metabolic, penetration, extraction, and sequestration, altered behaviour and cross-resistance.
Target site resistance often evolves from a genetic modification affecting a channel protein or receptor protein that is important to pest survival and also used as an insecticide binding site. In a normal situation an insecticide will attach to that protein at a target site and block its function, leading to invertebrate death. Mutation can lead to a target site that is a different shape to the usual configuration, making it more difficult for the insecticide to bind to the protein and block the function. When this happens, the interaction of the insecticide with its target site is impaired and the insecticide loses its efficacy. The effect of target site resistance results in an on/off scenario. If a pest carries target site resistance to a particular insecticide it will not be controlled with that insecticide.
Another common type of resistance, ‘metabolic resistance’, occurs when a pest already has some detoxifying enzymes that will degrade the insecticide and it builds up a higher number of those enzymes. Thus, this is using a mechanism that is already naturally at play within the pest and it can result in variable levels of resistance. Therefore, it can be difficult to predict the outcome of using an insecticide on a pest that has metabolic resistance.
When a pest becomes resistant to one insecticide (eg. dimethoate), the resistance can often extend to other chemically-related insecticides (eg. omethoate and chlorpyrifos); these related insecticides are labelled with a common Mode of Action (MoA) group number (eg. Group 1B, the organophosphates). This is because insecticides within a common MoA target a common site within the pest. In this case, dimethoate, omethoate, and chlorpyrifos all act by binding to, and blocking the activity of an important nervous system enzyme in invertebrates termed ‘acetylcholinesterase’.
Because all insecticides within a group share a common MoA, there is a high risk of cross-resistance to many insecticides in the same group. Although less common, cross-resistance can also occur across two (or more) MoA groups.
Table 1. Resistance occurrence and insecticide groups involved in key Australian grain pests.
The resistant pests of the southern grains region
Redlegged earth mites (RLEM)
Redlegged earth mite (Halotydeus destructor) RLEM is widespread in the southern cropping region and is a major and common pest of pastures and grain crops, particularly during seedling establishment when crop growth is most vulnerable, resulting in the potential for considerable economic losses. The economic impact varies across years. The mites have a very broad host range which includes canola, wheat, barley, oats, lupins, sunflower, faba beans, field peas, poppies, lucerne and vetch, as well as pasture legumes and grasses. While RLEM are less of a concern in cereal crops and in some pulses (eg. lentils and chickpeas), they do cause some damage.
There are approximately 200 insecticide products registered in Australia for RLEM, but these are primarily from three chemical groups – organophosphates (Group 1B, eg. dimethoate), pyrethroids (Group 3A, eg. alpha-cypermethrin) and neonicotinoids (Group 4A, eg. imidacloprid), very much narrowing the options to rotate MoA for managing resistance.
There are continuing risk factors influencing the ability to minimise evolution of resistance. The vast majority of canola crops are sown with insecticide-treated seed (mainly imidacloprid) applied prior to sale; in many instances growers are not offered an alternative. Insecticide seed dressings have also become more widely used in wheat, oats and barley, as well as on pastures. Most perennial pastures receive relatively few insecticide applications except TimeRite® directed applications in spring, which can be applied prior to the cropping phase.
Selection for resistance can result from targeted or non-targeted applications. That is, applications are either specifically targeted to control RLEM, or broad-spectrum products are applied to control a variety of pests at seedling establishment. Towards the end of the season, pyrethroids applied at the mature crop growth stages typically target caterpillars and aphids but invariably affect RLEM if applied before the diapause stage commences. In pastures and pulse crops, Lucerne flea is often a co-target, resulting in combination (tank mix) or repeat applications and more prominent use of organophosphates that target both mites and fleas. Tank mixes are not advised due to a lack of information about the efficacy of mixed products and the need to rotate chemicals between subsequent generations.
The complexity of dealing with one pest while influencing another can result in unintentional resistance evolution and needs to be considered with all insecticide applications.
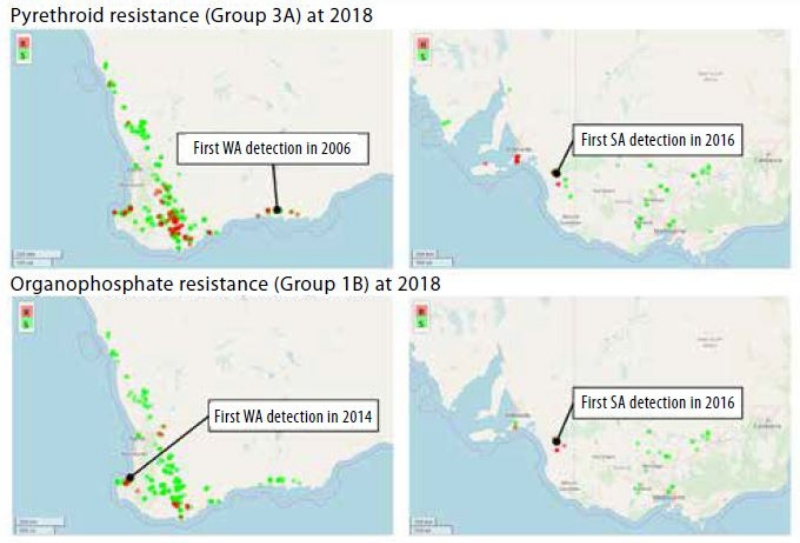
The RLEM resistance evolution is currently playing out in Western Australia and South Australia as shown in Figure 1.
Repeated insecticide applications of the same MoA group within and between seasons has led to the relatively rapid evolution and spread of resistance, which now includes the two main chemical groups: synthetic pyrethroids (synthetic pyrethroids, Group 3A) and organophosphates (organophosphates, Group 1B). Both are routinely applied against this pest in both the grains and grazing industries.
Figure 1. Distribution of insecticide resistance in RLEM. Source: cesar.
In South Australia, high levels of resistance to synthetic pyrethroids and moderate levels of resistance to organophosphates have been recorded. At the time of publication, insecticide resistance has not been detected in other parts of SA, Victoria, New South Wales or Tasmania. In the short-medium term, resistance is likely to become more prevalent in SA, and is expected to arise in other states. It is notable that at the time of writing, resistance profiling of RLEM populations is continuing through GRDC investment and a collaboration between cesar, the Western Australian Department of Primary Industries and Regional Development, and CSIRO. These activities will help us continue to build a picture about the current status of insecticide resistance in the grains industry.
Green peach aphid (GPA)
In Australia, the green peach aphid (GPA), Myzus persicae, primarily attacks canola and pulse crops across all grain growing regions, typically occurring every year or in some locations, every two to three years. Canola is more vulnerable to GPA attack than pulse crops. They are also a major pest of horticulture (hosts include a variety of vegetables and some fruit tree crops).
The aphids feed by sucking sap from leaves and flower buds. In grain crops, GPA typically cause less direct feeding damage than other aphid species, but it is their status as a disease vector that poses a significant threat. Nonetheless, when populations become large all foliage of a crop plant may be covered, resulting in retarded growth of young plants, however this is uncommon.
Young vegetative canola is most susceptible to GPA damage during autumn when GPA increase in numbers in the milder conditions and aphid flights are common. Large infestations of GPA on seedling crops can cause leaf distortion, wilting of cotyledons, stunting of growth, premature leaf senescence and seedling death. Although GPA may be found in canola at later stages, their numbers are usually insufficient to cause significant yield loss.
The aphids’ capacity to transmit virus to early establishing crops is an ongoing challenge for the industry. GPA can transmit more than 100 plant viruses, such as turnip yellows virus (TuYV) (previously known as beet western yellows virus: BWYV) and cucumber mosaic virus (CMV). Aphids need to feed on the plant to acquire TuYV as well as to transmit the virus. Hence, correctly timed insecticide application can prevent virus transmission.
Aphids produce offspring that are clones of the mother. For this reason, new GPA resistances can appear widespread in a single season and may occur across Australia over a couple of seasons.
Five chemical subgroups are registered to control GPA in grain crops: carbamates (Group 1A, eg. pirimicarb); synthetic pyrethroids (Group 3A, eg. alpha-cypermethrin); organophosphates (Group 1B, eg. dimethoate); neonicotinoids (Group 4A, eg., imidacloprid); and sulfoxaflor (Group 4C). Paraffinic spray oils are also registered for suppression of GPA.
Most canola crops are sown with imidacloprid-treated seed. Only a small proportion of pulse crops are sown with imidacloprid-treated seed.
During 2014, agronomists from all states were surveyed by cesar on their management of GPA, which provided the following insights:
- Foliar insecticides were sprayed in about 80% of canola paddocks annually, although many of these applications did not specifically target GPA.
- When insecticides were being applied for GPA, approximately one-third of sprays were used prophylactically.
- Except for seed treatments, pyrethroids, carbamates and organophosphates products were almost exclusively used to combat GPA in grains crops, with sulfloxaflor (Transform®) expected to have greater usage following its registration in 2013.
While seasonal challenges may vary chemical usage, particularly the likelihood of prophylactic spraying, the results of this survey provide the industry with some benchmarking.
GPA has evolved resistance to many insecticides globally (over 70 actives across a range of MoA groups). Within Australia, high levels of resistance to carbamates and pyrethroids are now widespread, with populations displaying target site resistance. Widespread moderate levels of resistance to organophosphates have been detected, as well as widespread low levels of neonicotinoid resistance. In these cases, populations display metabolic resistance.
Currently, there is no evidence of any resistance to sulfoxaflor in Australia. However, populations identified overseas that carry resistance mutations represent a biosecurity risk to Australia as they are not found here. An economic analysis undertaken by CSIRO in 2015 estimated that an incursion of dual imidacloprid/sulfoxaflor resistant GPA could lead to economic impact of more than AUD500 million based on additional management costs in canola and yield losses.
GPA populations move readily between grains and horticultural crops especially in regions where both industries operate. In horticulture, application rates of insecticides are much higher than in grains placing extra selection for resistance evolution.
Ultimately, the use of chemicals to control GPA in oilseed, pulses and horticulture crops continues to grow in Australia, placing strong selection pressure on the evolution of resistance in GPA. As aphids produce cloned offspring, resistant individuals can soon dominate a landscape if there is widespread use of the same insecticide group across paddocks and farms. The grains industry Resistance Management Strategy for GPA considers grain grower proximity to horticultural regions and gives advice accordingly.
Diamondback moth (DBM)
The diamondback moth (Plutella xylostella, DBM) is a pest of canola, mustard, Brassica vegetables and forage crops. DBM larvae feed on plant foliage, stems, flower heads and pods. The larvae can be found at any stage of canola development, with their numbers often increasing in the lead-up to flowering. Canola can tolerate considerable leaf damage before causing yield loss but feeding on developing flower buds can be economically damaging. Severe infestations can cause complete defoliation and yield losses of up to 80 per cent in canola.
Chemicals remain central to DBM control in canola and in the forage and vegetable brassica industries. Five chemical sub-groups are registered to control DBM in Australian canola crops: synthetic pyrethroids, organophosphates, spinosyns, avermectins, and Bacillus thuringiensis. There are approximately 170 insecticide products registered in Australia for DBM control, but these are primarily from the organophosphates and synthetic pyrethroids. Four newer insecticides with different MoAs are registered for DBM control in brassica vegetable crops.
The use of chemicals in canola and vegetable crops continues to grow in Australia, placing strong selection pressure on the evolution of resistance. DBM has a high propensity to evolve resistance and there are more than 82 insecticide compounds recorded globally to which DBM has evolved resistance.
In Australia, there is high and widespread levels of resistance to pyrethroids and organophosphates (which are generally ineffective), and low levels of resistance to avermectins and spinosyns and moderate levels of resistance to diamides in DBM in canola production regions. Higher levels of resistance to the avermectins, spinosyns and diamides occur in DBM in vegetable production regions.
Because DBM moths can disperse over large distances, resistant individuals can soon dominate a landscape if there is widespread use of the same insecticide group. The movement of DBM between industries and crops including canola (grains), vegetables (horticulture) and forage brassicas (grazing industries) increases the pressure for resistance selection in any one industry. Resistance management is further complicated because, for each of these industries, there is a great disparity in the number of registered MoAs.
There are only two synthetic insecticides (spinetoram and emamectin benzoate) and several Bacillus thuringiensis var. kurstaki products currently registered for use in canola and capable of reliably providing efficacious control of DBM. Even so, resistance to these synthetic MoAs has also been recorded occasionally.
Cotton bollworm (also known as corn earworm) Helicoverpa armigera
In south-eastern Australia, the cotton bollworm (Helicoverpa armigera) is only an occasional pest of pulses, oilseeds and winter cereals. In comparison to the better known and closely related native budworm (H. punctigera), it is responsible for fewer crop issues. Nonetheless it represents a significant challenge for the grains industry given the ongoing reliance on chemical control methods and its resistance status. In addition, the incidence of the pest appears to be gradually increasing in some areas, according to anecdotal information.
Larvae feed directly on flowers, pods and seed heads, for which there are economic thresholds. For high value consumption pulses, grain quality may also be downgraded at receivals through relatively low levels of damage. The species is most prevalent across all northern grain growing regions of eastern Australia and is usually in lower abundance in the south.
There are over 200 insecticide products registered in Australia against cotton bollworm for grain, cotton and vegetable crops. The majority of available insecticides are from three chemical groups with broad-spectrum activity: carbamates, organophosphates and synthetic pyrethroids. There are an additional three registered insecticide MoAs that are selective for Helicoverpa spp. and to which there is low or no resistance: emamectin benzoate, indoxacarb and diamides (chlorantraniliprole). These have become more widely used in pulses due to their high efficacy and relatively low impact on beneficial insects.
Cotton bollworm has evolved some level of resistance to the three chemical groups with broad spectrum activity (organophosphates, carbamates and pyrethroids).
Historically, it is this resistance that caused devastation in the cotton industry prior to the introduction of GM cotton. The use of chemicals to target H. armigera in grain crops continues to grow in Australia, placing strong selection pressure for the evolution of resistance in some of the more selective products.
There have been resistances detected (low to very low incidence) in other registered MoAs including spinosyns, Bt, Indoxacarb and the diamides. The fact that they are present is a concern, even though they are not yet resulting in widespread chemical control failures.
Managing insecticide resistance
Sustainable pest management strategies are required for the stewardship of newer and older chemistries used in grains. Successful resistance management, in Australia and internationally, has been based on the implementation of Insecticide Resistance Management Strategies (IRMS) and central to these, the application of Integrated Pest Management (IPM) tactics and the rotation of MoA groups. Greater adoption of IPM tactics, including the careful management of beneficials, cultural practices and monitoring, along with chemical rotation, can effectively reduce selection pressures on currently applied chemistries.
Effective and sustainable insecticide management aims to minimise the selection pressure on invertebrates to evolve insecticide resistance. By rotating chemicals from groups with different modes of action, successive generations of the pest are not repeatedly treated with the same chemical compound. This particularly applies to pests with multiple generations in the one season that may require several spray applications.
Selection for resistance can occur as a result of repeated applications of the same MoA group against that pest, or if the pest is present when the MoA group is used against other pests in the same crop/paddock (called non-target exposure). Table 2 presents a typical rotation from the southern region and highlights the non-target sprays to which typical crop pests are exposed. The most effective method of reducing this non-target selection is to:
- rotate chemicals used across a crop, and
- reduce the need for insecticides where possible through IPM practices.
In broadacre farming, rotating use of the commonly used synthetic pyrethroid (group 3A) and organophosphate groups (1B) with other groups (where possible), will help to minimise evolution of resistance in target and non-target pests.
Table 2. A typical rotation, with some possible pest and insecticide scenarios, and prospects for non-target resistance selection.
References
Back Pocket Guide – beneficial insects. GRDC (2012).
I SPY Manual – Insects of Southern Australian Broadacre Farming Systems Identification Manual and Education Resource © 2018.
Pests and Beneficials in Australian Cotton Landscapes Cotton Info (2016). www.cottoninfo.com.au
Resistance Management Strategies (RMS) for pests in Australian grains:
• RMS for green peach aphid and a report on the science that informed the RMS
• RMS for redlegged earth mite and a report on the science that informed the RMS
• RMS for diamondback moth in Australian canola and a report on the science that informed the RMS
• RMS for Helicoverpa armigera in Australian grains and a report on the science that informed the RMS.
Acknowledgements
This research was funded by the GRDC as part of the ‘Supporting the sustainable use of insecticides and local on-farm implementation of integrated pest management strategies in the GRDC southern region’ project.